Замечания:1 创始人: Site Editor Publish Time: 2026-04-26 Origin: Веб - сайт








Solid-state batteries (SSBs) are an emerging energy storage technology with high energy density and safety. Achieving SSB requires advancements in material discovery and processing. Currently, it is still challenging to fabricate SSB using ceramic ceramics. From a material processing perspective, polymer electrolytes may be a solution for manufacturing SSBs due to their flexibility, roll-to-roll processing, and excellent interfacial properties. Therefore, researchers need to design next-generation lightweight, flexible, solvent-free, and electrochemically stable polymer media materials with ultrafast and well-defined ion transport properties. Customizing the interdependent properties of nanostructures associated with ion transport is an installable method for predictively designing polymeric media with ultra-high conductivity. Ion transport is defined by three basic transport parameters: ion mobility, free ion concentration, and migration number. Among the different media types, polymer composite media have performance advantages that are commensurate with the two phases. Ceramic oxide phases have high conductivity and oxidation dendritics, while polymer phases, although less conductive, provide flexible and easily processable matrices for dispersing ceramic phases and synthesizing independent thin film pores with excellent interfacial properties with anodes and anodes. Currently, the structure-properties between ceramic phases and ion transport mechanisms support each other remains an interesting concept in SSB composite polymer electrolytes.
In view of this, Dr. Jianlin Li of Oak Ridge National Laboratory in the United States led a team to publish the latest research results entitled "Nanoscale Ion Transport Enhances Conductivity in Solid Polymer-Ceramic Lithium Electrolytes" in ACS Nano.
【Key Points】

Figure 1. SEM image of the electrospun Al-LLZO platelets. The scale bar is 10 µm. The inset shows a cross-sectional SEM image of a composite PEO electrolyte filled with 15 wt% Al-LLZO. The scale bar is 4 µm.
1. In this work, the authors established correlations between composite structure, polymer segment dynamics, and lithium-ion (Li+) transport in ceramic polymer composites.
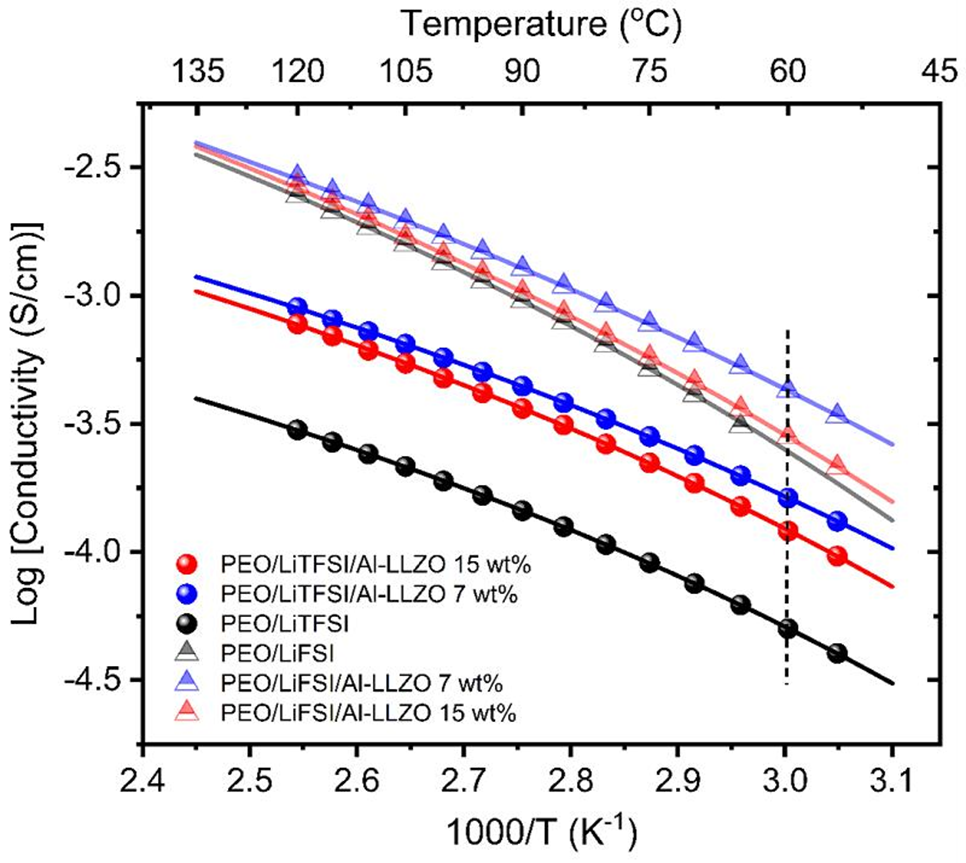
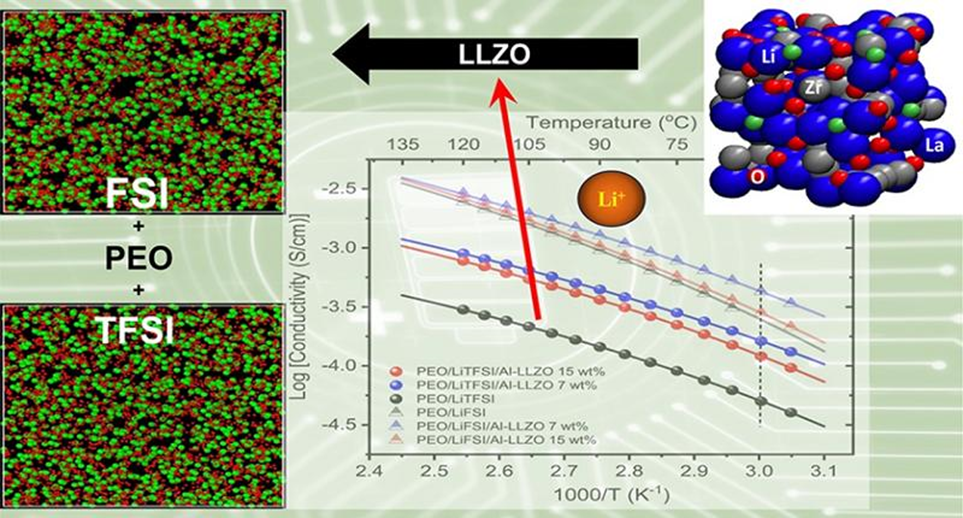
Figure 2. Summarized Arrhenius plot for the composite LiTFSI and LiFSI PEO electrolytes filled with Al-LLZO. The electrochemical testing was performed at 60oC (dotted line on the plot).
2. Elucidating this structure-performance relationship will allow the Li+ conductivity to be adjusted by optimizing the macroelectrochemical stability of the electrolyte. The authors found that ion dissociation of slow polymer segment dynamics can be enhanced by controlling the morphology and function of the polymer/ceramic interface. The chemical structure of Li+ salts in composite electrolytes is related to the size of the ion cluster domain, the conductive mechanism and the electrochemical stability of the electrolyte.
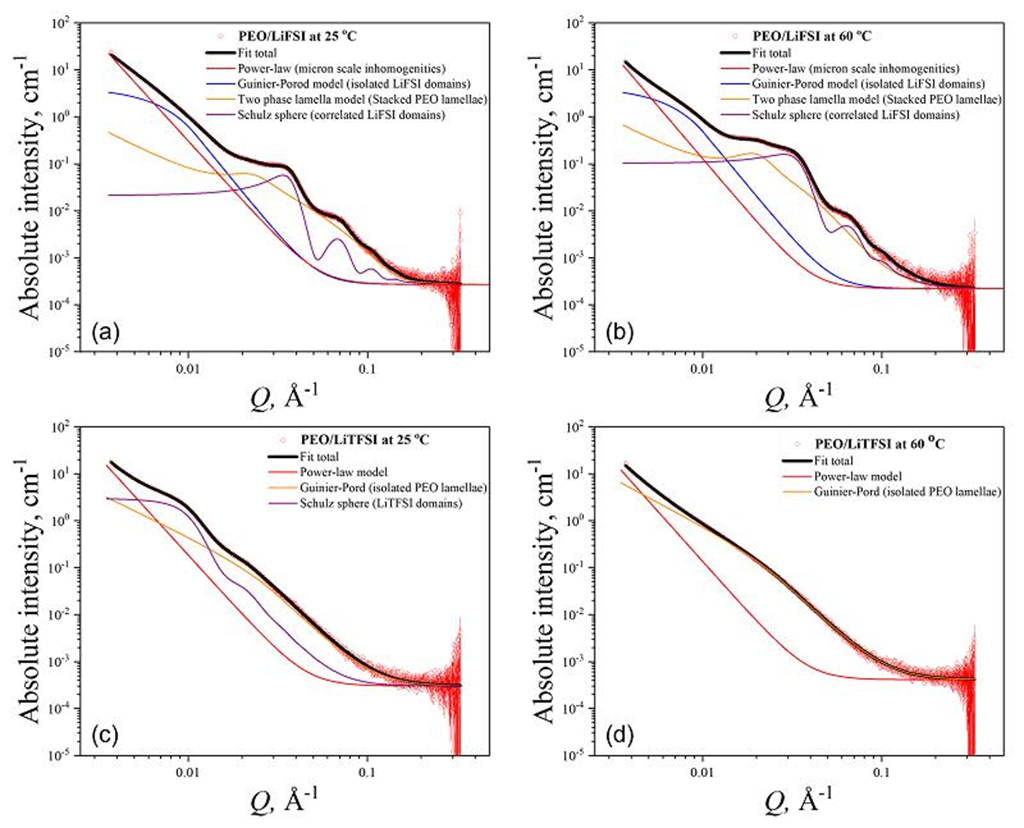
Figure 4. Experimental SAXS patterns and model-fits of the (a, b) PEO/LiFSI electrolytes at 25oCand60oC. (c, d) PEO/LiTFSI electrolytes at 25oC and 60oC. The SAXS model-fits were based on multiple SAXS model functions as indicated in each plot. The fitting parameters of the SAXS functions that were used to fit the scattering curves are summarized in the Supporting Information.
3. The authors used polyethylene oxide (PEO) filled with bis(trifluoromethanesulfonyl)lithium imide (LiTFSI) or bis(fluorosulfonyl)lithium imide (LiFSI) salts as the matrix. In addition, garnet electrolytes with planar geometries, aluminum-substituted lithium lanthanum zirconium oxide (Al-LLZO) are used in the ceramic nanoparticle section.
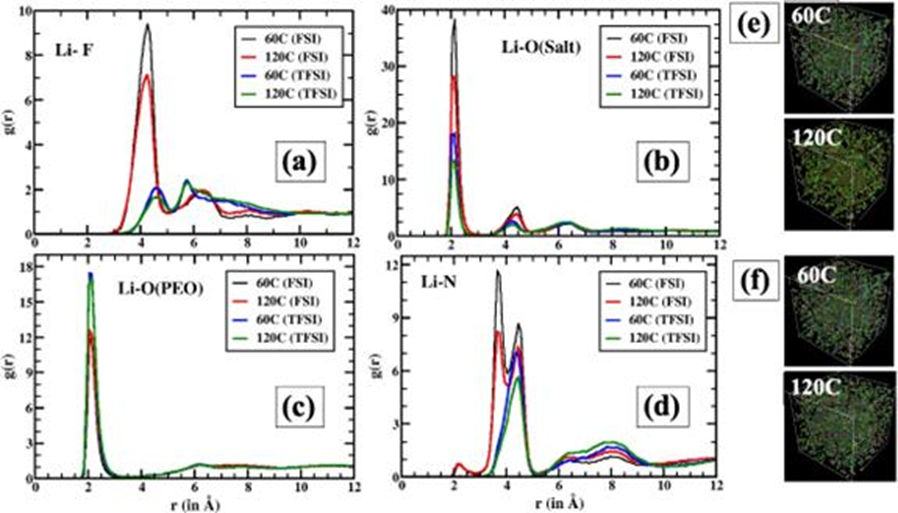
Figure 5. Structural behavior of Li+ ions. Radial distribution function (RDF) of Li+ with respect to (a) fluorine, (b-c) oxygen of salt and oxygen of PEO at two different temperatures, 60 ºC and 120 ºC. (d) Li+ with nitrogen atoms of salt. (e) and (f) snapshots showing LiFSI and LiTFSI respectively. For clarity only Li+ and Li+ salts are shown.
4. The authors investigated the kinetics of strong binding and highly fluid Li+ using dielectric relaxation spectroscopy. The incorporation of Al-LLZO wafers increases the quantitative density of the more mobile Li+.
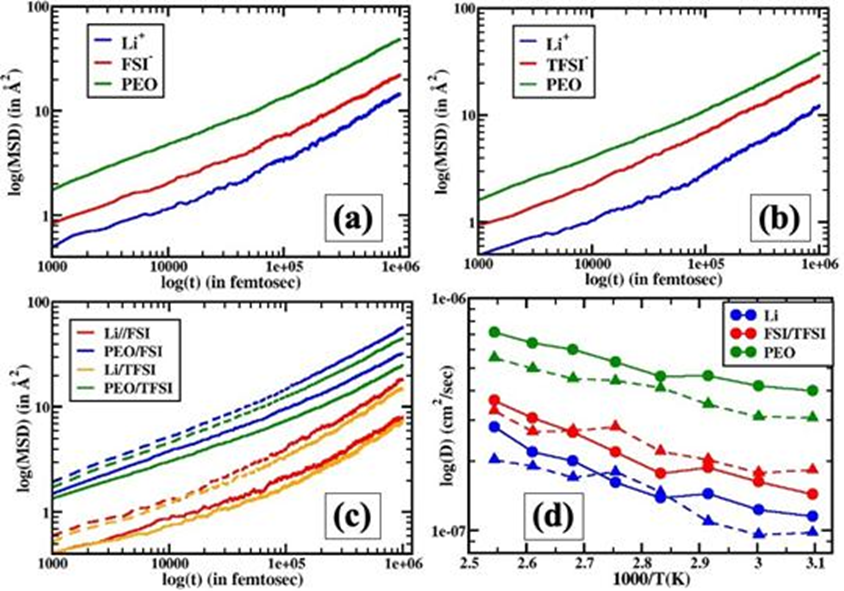
Figure 6. The mMean- square- displacement (MSD) and diffusivitiesy of Lithe +⁺, FSI⁻/TFSI⁻ anions, and PEO chains. (a) Comparison of MSDs for Li+⁺, FSI⁻, and PEO MSDs forin LiFSI samples at 50 º °C. (b) Comparison of MSDs foor Li+⁺, TFSI⁻, and PEO MSDs forin LiTFSI samples at 50 º °C. (c) Comparison of Li+⁺ and PEO dynamics (MSDs) for LiFSI and LiTFSI at 50 ºC (solid lines) and 120 º °C (dashed lines). The color schemes are respectivelyshowindicated in the legends. (d) Diffusivity of Li, calculated from Einstein’s relation,+⁺, FSI⁻/TFSI⁻, and PEO chain.s calculated Tusing the cEinstein relation. Circles (solid lines) and triangles (dashed lines) represent LiFSI and LiTFSI samples, respectively.
5. The authors studied the nanoscale ion aggregation structure by small-angle X-ray scattering, while simultaneously conducting molecular dynamics (MD) simulation studies to obtain the basic mechanism of Li+ de-correlation with long PEO chains in LiTFSI and LiFSI salts.
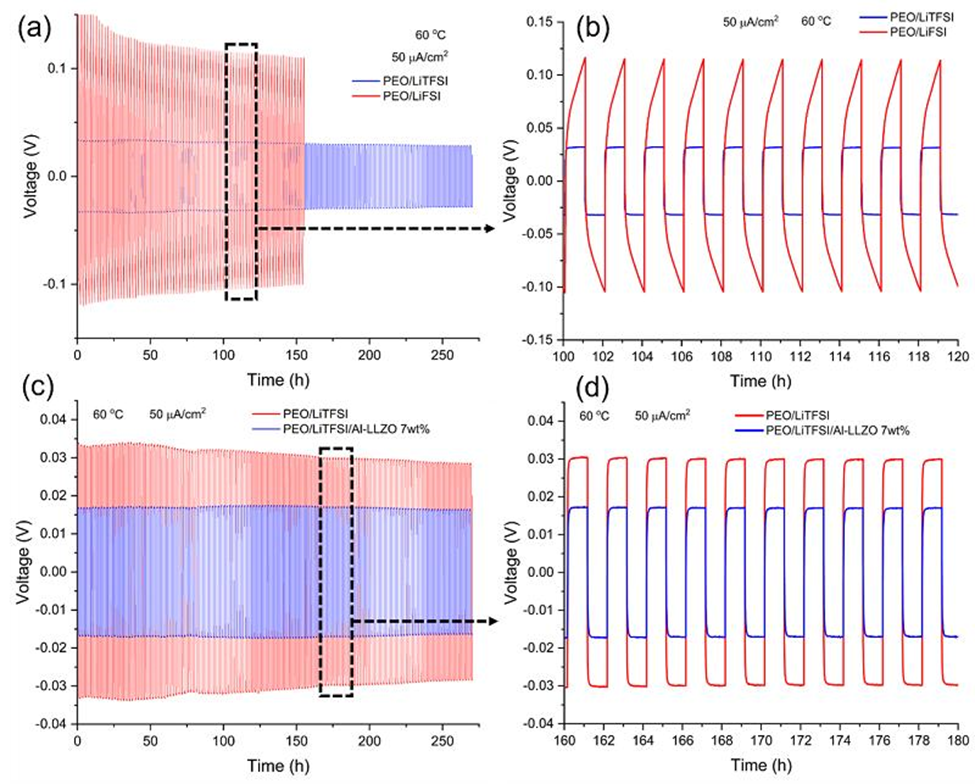
Figure 7. Comparison of the long term galvanostatic cycling of the (a, b) LiFSI and LiTFSI electrolytes and (c, d) LiTFSI and LiTFSI composite filled with 7 wt% Al-LLZO at 60 oC and 50 µA/ cm-2.
 Безопасность сети 32058300438
Безопасность сети 32058300438